Dynamic Organization of the Plant Cell
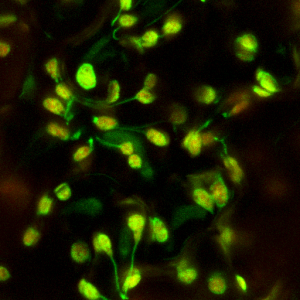
The ability to label different subcellular locations with the green fluorescent protein (GFP) has made it possible to visualize intracellular activities in living cells. We have introduced chimeric genes which express GFP in a variety of organelles within the plant cell in order to study the dynamics of cell organization. Labeling plastids with GFP led to the rediscovery of tubules emanating from plastids. We named these stromules, for stroma-filled tubules, the function and mechanism of formation of these unexpected structures remains a topic for study.
Click here to view our web essay on stromules.
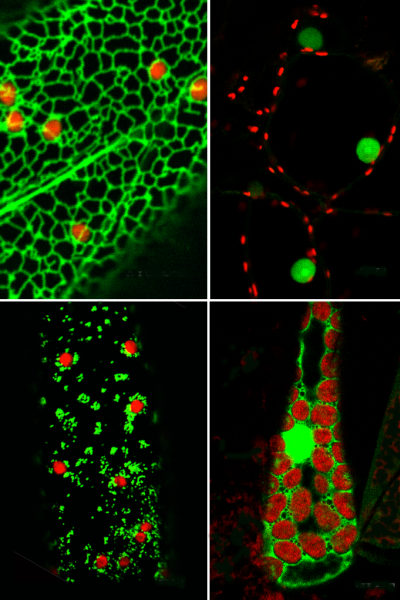
We have investigated whether myosins are involved in movement of stromules, plastids, mitochondria, peroxisomes, and other subcellular structures. Plant genomes encode the myosin XI family of myosin proteins. We have placed fluorescent labels on portions of the tail regions of Arabidopsis myosin XIs in order to determine where they localize, and therefore what role a particular myosin may play in movement of different types of organelles.
See a recent review and a previous commentary for more information about stromules.
Project Sponsors
The Chemical Sciences, Geosciences and Biosciences Division,
Office of Basic Energy Sciences, Office of Science, U.S. Department of Energy

